From single cells to tissues: Interactions between the matrix and human breast cells in real time
PLoS ONE
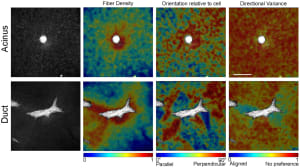
Bulk properties do not determine shape; however, localized regions of collagen fiber alignment are required for ductal elongation and branching.
Abstract
Background: Mammary gland morphogenesis involves ductal elongation, branching, and budding. All of these processes are mediated by stroma - epithelium interactions. Biomechanical factors, such as matrix stiffness, have been established as important factors in these interactions. For example, epithelial cells fail to form normal acinar structures in vitro in 3D gels that exceed the stiffness of a normal mammary gland. Additionally, heterogeneity in the spatial distribution of acini and ducts within individual collagen gels suggests that local organization of the matrix may guide morphogenesis. Here, we quantified the effects of both bulk material stiffness and local collagen fiber arrangement on epithelial morphogenesis.
Results: The formation of ducts and acini from single cells and the reorganization of the collagen fiber network were quantified using time-lapse confocal microscopy. MCF10A cells organized the surrounding collagen fibers during the first twelve hours after seeding. Collagen fiber density and alignment relative to the epithelial surface significantly increased within the first twelve hours and were a major influence in the shaping of the mammary epithelium. The addition of Matrigel to the collagen fiber network impaired cell-mediated reorganization of the matrix and increased the probability of spheroidal acini rather than branching ducts. The mechanical anisotropy created by regions of highly aligned collagen fibers facilitated elongation and branching, which was significantly correlated with fiber organization. In contrast, changes in bulk stiffness were not a strong predictor of this epithelial morphology.
Conclusions: Localized regions of collagen fiber alignment are required for ductal elongation and branching suggesting the importance of local mechanical anisotropy in mammary epithelial morphogenesis. Similar principles may govern the morphology of branching and budding in other tissues and organs.
Keywords: Collagens, Morphogenesis, Extracellular matrix, Gels, Anisotropy, Stiffness, Scanning electron microscopy, Mammary gland development




